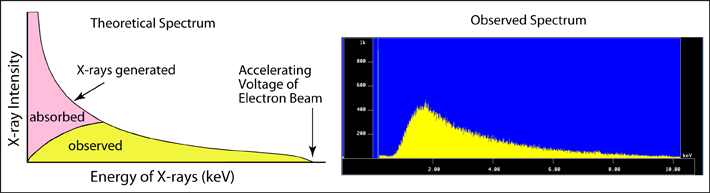
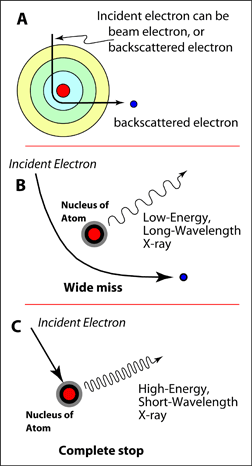
Continuum X-rays are produced by the interaction of either beam electrons or backscattered electrons with the Coulombic field associated with the nucleus of an atom. As an electron passes close to the nucleus, it may be deflected and lose some of its energy. This energy will be given off in the form of an X-ray. How much energy is given off will be determined by the nature of the interaction. If the distance between the nucleus of the atom and the electron is large, then the electron will lose only a small amount of its energy, and the X-ray given off will be low-energy. However, if the electron comes to a complete stop, it will produce a high-energy X-ray. The maximum amount of energy that the X-ray could have would be equal to the accelerating voltage being used by the instrument. The result is that there will be a continuum between virtually no energy lost to total energy loss, or very low-energy X-rays to high-energy X-rays. A theoretical spectrum is shown below for these continuum X-rays. Most of the X-rays produced will be of a very low-energy nature, fewer of the high-energy types will be produced. However the very low-energy X-rays will also be preferentially absorbed by the sample itself. Therefore most of these will never leave the sample. As a result, the observed continuum spectrum has a characteristic rise between about 1 and 3 keV. The intensity of the continuum X-rays will depend on the average atomic number of the area. A sample with a higher average atomic number will produce more interactions between the electrons and the nuclei of its atoms, and therefore more continuum X-rays will be produced. Because the continuum spectrum will increase or decrease with the composition of the material, it is important that during an analysis this background spectrum gets subtracted from the intensity of the characteristic peak in order to get its true intensity. |
 |